[ad_1]
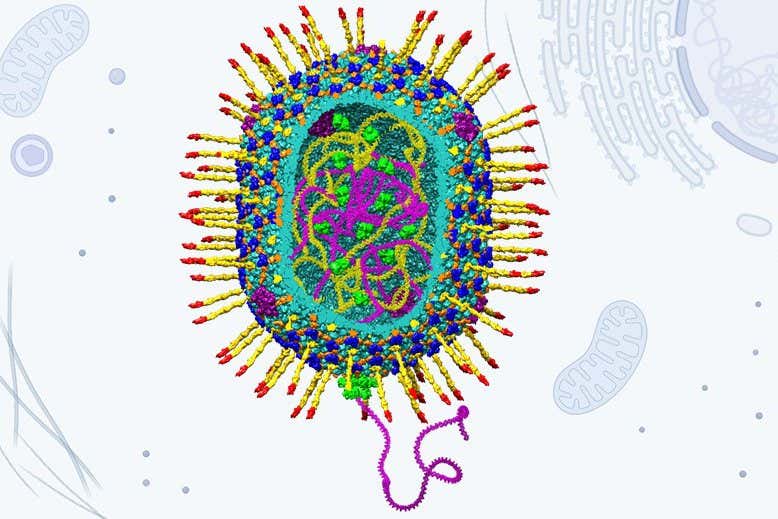
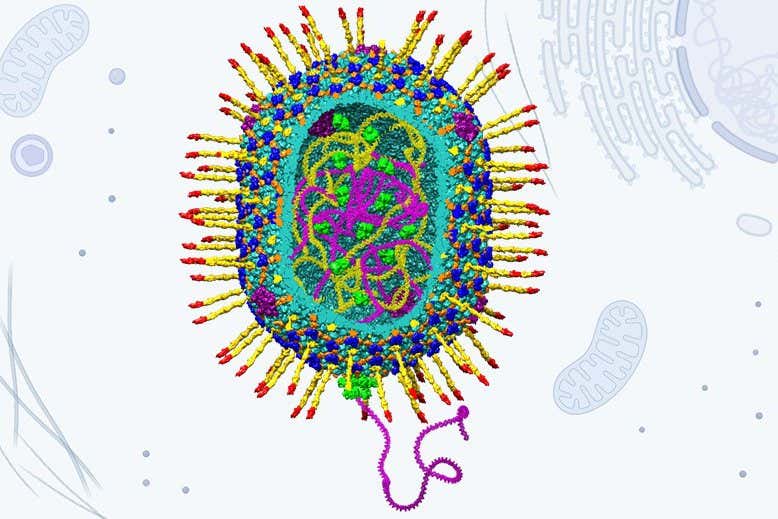
Model of bacteriophage T4, which the gene therapy-advancing virus is based on
Venigalla B. Rao; Victor Padilla-Sanchez, Andrei Fokine, and Jingen Zhu
A modified bacteria-killing virus, known as a phage, can deliver far more DNA to human cells than is possible with existing techniques. This capacity could lead to major advances in cell and gene therapies by allowing more sophisticated changes to be made to cells in a single step.
The modified virus can carry DNA strands up to 171,000 base pairs long – around 20 times as much as the largest existing viruses used for gene therapies. In addition to this DNA, it can carry more than 1000 other molecules, such as RNAs and proteins, says Venigalla Rao at The Catholic University of America in Washington DC.
“We can combine all of these in one particle and be able to aim not only for therapy, but potentially for a cure,” says Rao.
A growing number of treatments involve modifying cells inside or outside of the body, but delivering the necessary components to cells remains a huge challenge.
For instance, some people have a condition that causes progressive muscle weakness, called Duchenne muscular dystrophy, that is due to mutations in a gene for a protein called dystrophin. Efforts to develop gene therapies for the condition have been stymied by the fact that DNA of around 11,000 base pairs long is required to make the full-size dystrophin protein – more than fits in existing viruses.
In one experiment, Rao’s team delivered a dystrophin gene to human cells growing in culture and showed that the cells produced the full-size protein.
In another experiment, the team delivered multiple molecules to human cells at once, allowing them to edit several genes, switch off other genes and get each cell to produce various proteins, all at the same time.
The modified delivery virus is based on a T4 bacteriophage that usually infects only specific kinds of bacteria. Thanks to studies by Rao’s team and other research groups, the T4 virus is so well understood that it can be substantially altered and customised.
In particular, Rao’s team added a coating that results in the virus being engulfed by human cells and in this way gets its cargo inside them.
These modified viruses will also be much easier and cheaper to manufacture than the viruses currently used for gene therapy, says Rao, as they don’t need to be grown in human cells.
However, Rao and his colleagues haven’t yet demonstrated that the viruses can be used to deliver genes to cells in bodies, says Jeffrey Chamberlain at the University of Washington in Seattle, whose team is trying to develop gene therapies for Duchenne muscular dystrophy by splitting the gene between several viruses.
“Nonetheless, the early data are encouraging, and it will be interesting to follow further developments,” says Chamberlain. There is a great need for additional systems that deliver gene therapies into various cells and organs in the body, he says.
It may take a lot of extra work to get the virus to work well in people’s bodies, Rao says, but he thinks it is feasible. More immediately, the modified virus could be used to alter cells outside the body for treating people.
For instance, some cancers are now treated by modifying immune cells to target tumours. This often involves several steps: first using a virus to deliver a targeting gene, then making additional changes by separately delivering gene-editing components. The result is a mix of cells that don’t all have the desired changes, which makes them less effective when injected into a person with cancer.
Being able to deliver the targeting gene and gene-editing components in a single virus would greatly improve the process.
Topics:
[ad_2]
Source link